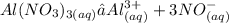Chemistry, 06.08.2021 22:30 kyramillerr8639
Which of the following has the greatest effect on colligative properties?
A. Calcium chloride (CaCl2)
B. Sodium chloride (NaCl)
C. Aluminum Nitrate (Al(NO3)3)
D. Epsom salt (MgSO4)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2


Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2
You know the right answer?
Which of the following has the greatest effect on colligative properties?
A. Calcium chloride (CaCl...
Questions


History, 07.11.2019 22:31

Mathematics, 07.11.2019 22:31

Biology, 07.11.2019 22:31

Spanish, 07.11.2019 22:31



Social Studies, 07.11.2019 22:31

English, 07.11.2019 22:31

English, 07.11.2019 22:31


Mathematics, 07.11.2019 22:31


Mathematics, 07.11.2019 22:31

Physics, 07.11.2019 22:31


Mathematics, 07.11.2019 22:31

History, 07.11.2019 22:31


Mathematics, 07.11.2019 22:31