
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the water (dehydration). A 43.19 gram sample of a hydrate of MgBr2 was heated thoroughly in a porcelain crucible, until its weight remained constant. After heating, 27.21 grams of the anhydrous compound remained. What is the formula of the hydrate?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 2

Chemistry, 23.06.2019 07:00
Scuba divers use tanks of compressed air to them breathe. gases can be compressed because?
Answers: 1

You know the right answer?
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the w...
Questions

Chemistry, 09.12.2020 21:40


Advanced Placement (AP), 09.12.2020 21:40

Biology, 09.12.2020 21:40





Mathematics, 09.12.2020 21:40




Social Studies, 09.12.2020 21:40


Biology, 09.12.2020 21:40

Law, 09.12.2020 21:40

Mathematics, 09.12.2020 21:40



 .
. denote the number of
denote the number of 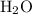 formula units for every
formula units for every  formula unit. The formula of the hydrate would be
formula unit. The formula of the hydrate would be  .
. :
: 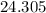 .
. :
: 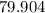 .
. :
:  .
. :
: 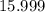 .
. .
. .
.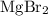 . There was
. There was 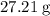 of this compound. Calculate the number of moles of formula units in that much of this compound:
of this compound. Calculate the number of moles of formula units in that much of this compound: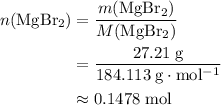 .
.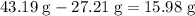 of water
of water 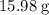 of
of 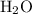 :
: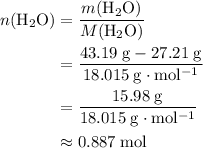 .
. of
of 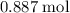 of
of 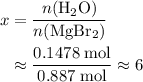 .
.

