Chemistry, 02.08.2021 22:50 gretchcampbell
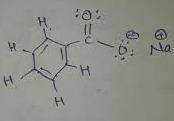
1. Draw the condensed structural formula of sodium benzoate showing all charges, atoms including any lone pairs in the side chain functional group, and all sigma and pi bonds.
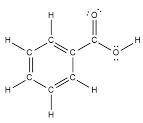
2. Draw the condensed structural formula of benzoic acid showing all atoms including any lone pairs in the side chain functional group, and all sigma and pi bonds. Indicate the acidic hydrogen.
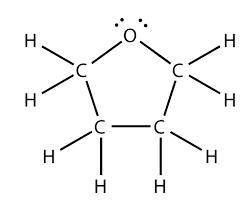
3. Draw the condensed structural formula of tetrahydrofuran (THF) showing all heteroatoms plus their lone pairs and all sigma and pi bonds.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2

Chemistry, 23.06.2019 14:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 6.00 mol fe and 8.45 mol nio(oh) react?
Answers: 1
You know the right answer?
1. Draw the condensed structural formula of sodium benzoate showing all charges, atoms including any...
Questions






English, 07.04.2021 17:00


Mathematics, 07.04.2021 17:00






History, 07.04.2021 17:00

Social Studies, 07.04.2021 17:00



Mathematics, 07.04.2021 17:00

Mathematics, 07.04.2021 17:00

Mathematics, 07.04.2021 17:00