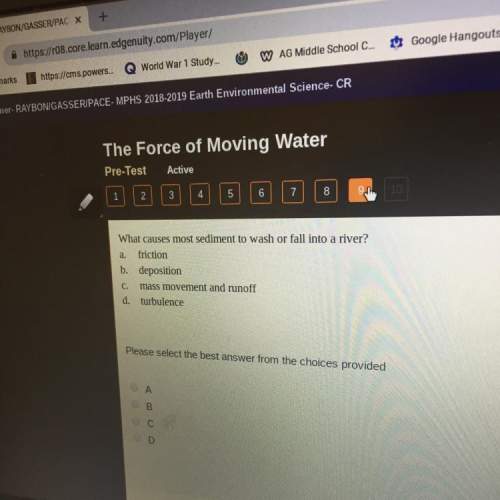
A 3.0 L cylinder is heated from an initial temperature of 273 K at a pressure of 105 kPa to a final temperature of 333 K. Assuming the amount of gas and the volume remain the same, what is the pressure (in kilopascals) of the cylinder after being heated?
pressure:
kPa

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Write the symbol for every chemical element that has atomic number greater than 3 and atomic mass less than 12.0 u.
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

You know the right answer?
A 3.0 L cylinder is heated from an initial temperature of 273 K at a pressure of 105 kPa to a final...
Questions

Mathematics, 29.05.2021 16:00

Mathematics, 29.05.2021 16:00


Computers and Technology, 29.05.2021 16:00



Computers and Technology, 29.05.2021 16:00


History, 29.05.2021 16:00

Advanced Placement (AP), 29.05.2021 16:00


Mathematics, 29.05.2021 16:00

History, 29.05.2021 16:00

Mathematics, 29.05.2021 16:00





Mathematics, 29.05.2021 16:00

Mathematics, 29.05.2021 16:00