Chemistry, 30.07.2021 02:10 bobelliot67789
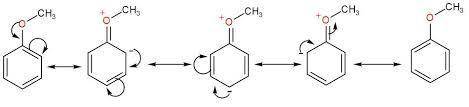
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Groups, already present on the benzene ring, that direct ortho/para further stabilize this intermediate by participating in the resonance delocalization of the positive charge. Assume that the following group is present on a benzene ring at position 1 and that you are brominating the ring at positon 4. Draw the structure of the resonance contributor that shows this group actively participating in the charge delocalization. OCH3

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which statement about sound is not true? a. air particles travel with sound waves. b. sound waves cannot travel through a vacuum. c. sound waves exist even if no one hears them. d. air particles vibrate along the path of a sound wave.
Answers: 1

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1
You know the right answer?
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Gr...
Questions

Biology, 24.08.2019 22:30

Biology, 24.08.2019 22:30

Chemistry, 24.08.2019 22:30

Mathematics, 24.08.2019 22:30


Mathematics, 24.08.2019 22:30

History, 24.08.2019 22:30

English, 24.08.2019 22:30


History, 24.08.2019 22:30




Spanish, 24.08.2019 22:30

World Languages, 24.08.2019 22:30

French, 24.08.2019 22:30

History, 24.08.2019 22:30

Social Studies, 24.08.2019 22:30


Biology, 24.08.2019 22:30