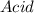In the following acid-base reaction,
H3O+ is the .
HCI(g) + H2O(l) +H30+ (aq) + Cl-(aq)
...

Chemistry, 27.07.2021 03:50 Irishstoner5608
In the following acid-base reaction,
H3O+ is the .
HCI(g) + H2O(l) +H30+ (aq) + Cl-(aq)
A acid
B conjugate acid
C conjugate base

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 09:30
How many moles of na2s2o3 are needed to react with 0.12mol of cl2? show work.
Answers: 1
You know the right answer?
Questions

Computers and Technology, 03.08.2019 04:10


Spanish, 03.08.2019 04:10

Spanish, 03.08.2019 04:10


Spanish, 03.08.2019 04:10



Social Studies, 03.08.2019 04:10





English, 03.08.2019 04:10




Mathematics, 03.08.2019 04:10