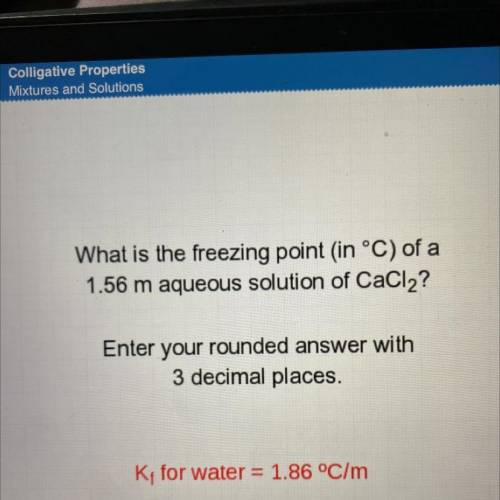
What is the freezing point in °C) of a
1.56 m aqueous solution of CaCl2?
Enter your rounded a...

Chemistry, 26.07.2021 22:20 joselinegarciaowyrpf
What is the freezing point in °C) of a
1.56 m aqueous solution of CaCl2?
Enter your rounded answer with
3 decimal places.
K; for water = 1.86 °C/m

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which function is performed by earths atmosphere? a. ultraviolet rays are prevented from reaching the ozone layer. b. earths temperature is raised and moderated by trapping in heat c. charged particles from the sun are prevented from reaching earth. d. magnetic charges from space are prevented from reaching earths surface.
Answers: 2

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2
You know the right answer?
Questions

Mathematics, 05.12.2019 21:31


Mathematics, 05.12.2019 21:31

English, 05.12.2019 21:31

Social Studies, 05.12.2019 21:31

Mathematics, 05.12.2019 21:31

Mathematics, 05.12.2019 21:31









Biology, 05.12.2019 21:31



Social Studies, 05.12.2019 21:31

English, 05.12.2019 21:31



