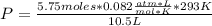A chemist is preparing to carry out a reaction that requires 5.75 moles of hydrogen gas. The chemist pumps the hydrogen into a 10.5 L rigid steel container at 20.0 °C. To what pressure, in kPa, must the hydrogen be compressed? (Show all work for full credit and circle your final answer) *

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 23.06.2019 04:31
Which of the following is an example of how telecommunication devices people do their jobs? a.) a security guard checks the time using a digital watch. b.) a banker does some quick math using a solar calculator. c.) a nurse uses a digital thermometer to take a patient’s temperature. d.) a construction worker reports in to his office using a cell phone.
Answers: 1
You know the right answer?
A chemist is preparing to carry out a reaction that requires 5.75 moles of hydrogen gas. The chemist...
Questions

Physics, 20.11.2020 04:30

Mathematics, 20.11.2020 04:30


Mathematics, 20.11.2020 04:30



Mathematics, 20.11.2020 04:30

Chemistry, 20.11.2020 04:30




English, 20.11.2020 04:30

Mathematics, 20.11.2020 04:30




Mathematics, 20.11.2020 04:30


Physics, 20.11.2020 04:30

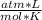 T= 20 C= 293 K (being 0 C= 273 K)
T= 20 C= 293 K (being 0 C= 273 K)