
Chemistry, 24.07.2021 21:00 mickimlvn6128
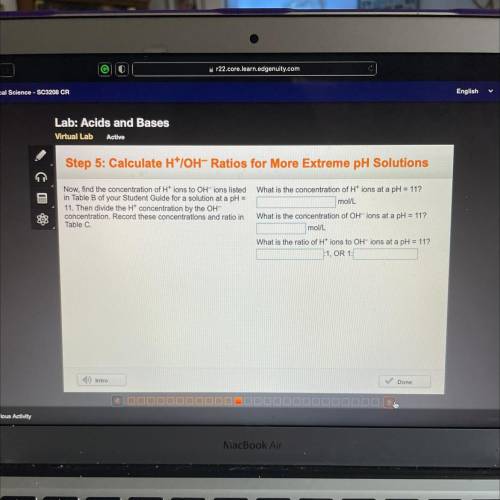
Step 5: Calculate H*IOH-Ratios for More Extreme pH Solutions
What is the concentration of H+ ions at a pH = 11?
mol/L
Now, find the concentration of H+ ions to OH-ions listed
in Table B of your Student Guide for a solution at a pH =
11. Then divide the Ht concentration by the OH-
concentration. Record these concentrations and ratio in
Table C.
What is the concentration of OH-ions at a pH = 11?
mol/L
What is the ratio of H+ ions to OH-ions at a pH = 11?
:1, OR 1:

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 00:00
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1
You know the right answer?
Step 5: Calculate H*IOH-Ratios for More Extreme pH Solutions
What is the concentration of H+ ions a...
Questions

Mathematics, 27.06.2019 06:50


History, 27.06.2019 06:50

History, 27.06.2019 06:50



Mathematics, 27.06.2019 06:50

Mathematics, 27.06.2019 06:50


Mathematics, 27.06.2019 06:50

Geography, 27.06.2019 06:50



Mathematics, 27.06.2019 06:50

Mathematics, 27.06.2019 06:50



Spanish, 27.06.2019 06:50




