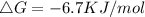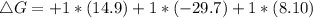Chemistry, 23.07.2021 01:00 yasiroarafat12
In nature, one common strategy to make thermodynamically unfavorable reactions proceed is to couple them chemically to reactions that are thermodynamically favorable. As long as the overall reaction is thermodynamically favorable, even the unfavorable reaction will proceed.
Part A
Consider these hypothetical chemical reactions:
A⇌B,ΔG= 14.8 kJ/mol
B⇌C,ΔG= -29.7 kJ/mol
C⇌D,ΔG= 8.10 kJ/mol
What is the free energy, ΔG, for the overall reaction, A⇌D?
Part B
Firefly luciferase is the enzyme that allows fireflies to illuminate their abdomens. Because this light generation is an ATP-requiring reaction, firefly luciferase can be used to test for the presence of ATP. In this way, luciferase can test for the presence of life. The coupled reactions are
luciferin+O2ATP⇌⇌oxyluciferin+light AMP+PPi
If the overall ΔG of the coupled reaction is -7.50 kJ/mol , what is the equilibrium constant, K, of the first reactions at 11 ∘C ? The ΔG for the hydrolysis of ATP to AMP is −31.6 kJ/mol.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

You know the right answer?
In nature, one common strategy to make thermodynamically unfavorable reactions proceed is to couple...
Questions



Mathematics, 30.08.2021 20:10



Mathematics, 30.08.2021 20:10



Geography, 30.08.2021 20:10

Biology, 30.08.2021 20:10



Mathematics, 30.08.2021 20:10

Mathematics, 30.08.2021 20:10




Mathematics, 30.08.2021 20:10


Chemistry, 30.08.2021 20:10