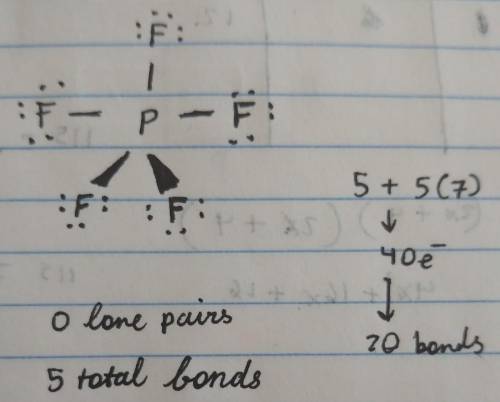Consider the molecule PF5.
Indicate how many lone pairs you would find on the central atom:
I...

Chemistry, 22.07.2021 22:50 vanessajimenez361
Consider the molecule PF5.
Indicate how many lone pairs you would find on the central atom:
Indicate how many total bonds are connected to the central atom (count single bonds as 1 bond, double bonds as 2 bonds, and triple bonds as 3 bonds):

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3


Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3
You know the right answer?
Questions

Chemistry, 24.09.2020 14:01


Biology, 24.09.2020 14:01


History, 24.09.2020 14:01

Mathematics, 24.09.2020 14:01






Chemistry, 24.09.2020 14:01


Mathematics, 24.09.2020 14:01

Chemistry, 24.09.2020 14:01

Geography, 24.09.2020 14:01

Biology, 24.09.2020 14:01