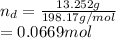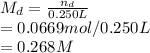Chemistry, 22.07.2021 22:40 lukecoupland4401
An intravenous solution was prepared by add-in 13.252 g of dextrose (C6H12O6) and 0.686 g of sodium chloride to a 250.0 mL volumetric flask and diluting to the calibration mark with water. What is the molarity of each component of the solution

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1


Chemistry, 23.06.2019 05:30
For the reaction i2(g)+br2(g)←−→2ibr(g), kc=280 at 150 ∘c. suppose that 0.450 mol ibr in a 2.00-l flask is allowed to reach equilibrium at 150 ∘c. what is the equilibrium concentration of 2ibr, i2, br2
Answers: 1
You know the right answer?
An intravenous solution was prepared by add-in 13.252 g of dextrose (C6H12O6) and 0.686 g of sodium...
Questions



Mathematics, 11.06.2021 08:20

English, 11.06.2021 08:20

Social Studies, 11.06.2021 08:20

History, 11.06.2021 08:20

Geography, 11.06.2021 08:20

World Languages, 11.06.2021 08:20

Mathematics, 11.06.2021 08:20



Mathematics, 11.06.2021 08:20




Physics, 11.06.2021 08:20

English, 11.06.2021 08:20


Biology, 11.06.2021 08:20

Mathematics, 11.06.2021 08:20

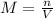
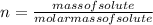
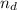 ) is:
) is: