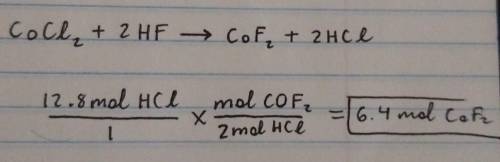Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2


Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
Calculate the amount of mole(s) of CoF2 required to react with 12.8 moles of HCI.
CoCl2 + 2HF -->...
Questions

History, 27.04.2021 05:40




Biology, 27.04.2021 05:40

English, 27.04.2021 05:40

Health, 27.04.2021 05:40

Mathematics, 27.04.2021 05:40

Physics, 27.04.2021 05:40








Mathematics, 27.04.2021 05:40


English, 27.04.2021 05:40