
Chemistry, 21.07.2021 01:00 QueenZenobia
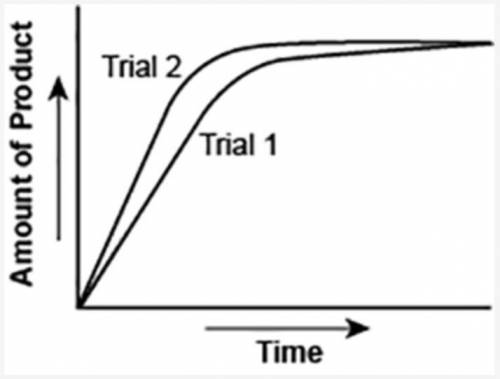
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
Which of the following statements explains which trial has a lower concentration of the reactant?
A: Trial 1, because the average rate of the reaction is lower.
B: Trial 1, because this reaction lasted for a longer duration than Trial 2.
C: Trial 2, because this reaction was initially fast and later slowed down.
D: Trial 2, because the volume of product formed per unit time was higher.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1
You know the right answer?
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different...
Questions

Mathematics, 30.07.2019 05:00









History, 30.07.2019 05:00

Biology, 30.07.2019 05:00

History, 30.07.2019 05:00

History, 30.07.2019 05:00

History, 30.07.2019 05:00

History, 30.07.2019 05:00




Biology, 30.07.2019 05:00

History, 30.07.2019 05:00



