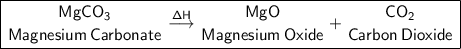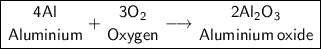Chemistry, 16.07.2021 06:00 rorathan123
Complete and balance the following chemical equations. Identify the reaction type as: combination, decomposition, single replacement, double replacement, or combustion.
Products:
Magnesium Oxide + Carbon dioxide.
a) MgCO₃ (Heat is supplied to the reaction (triangle over a arrow) -> Reaction type:
Products:
Aluminum Oxide
b) Al + O₂ -> Reaction type:

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1



You know the right answer?
Complete and balance the following chemical equations. Identify the reaction type as: combination, d...
Questions


Mathematics, 24.06.2019 21:10

English, 24.06.2019 21:10

Social Studies, 24.06.2019 21:10



English, 24.06.2019 21:10












English, 24.06.2019 21:10