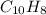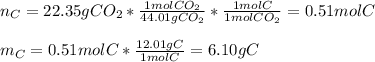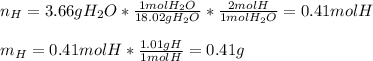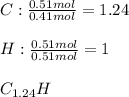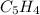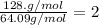Chemistry, 16.07.2021 02:30 ayoismeisjuam
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 128. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product carbon dioxide water mass 22.35 g 3.66 g Use this information to find the molecular formula of x

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1


Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2

Chemistry, 23.06.2019 02:00
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2
You know the right answer?
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions


Biology, 24.07.2019 12:00


Mathematics, 24.07.2019 12:00



Chemistry, 24.07.2019 12:00



Mathematics, 24.07.2019 12:00


History, 24.07.2019 12:00




History, 24.07.2019 12:00


Chemistry, 24.07.2019 12:00


Mathematics, 24.07.2019 12:00