
Chemistry, 16.07.2021 01:50 needthehelp78
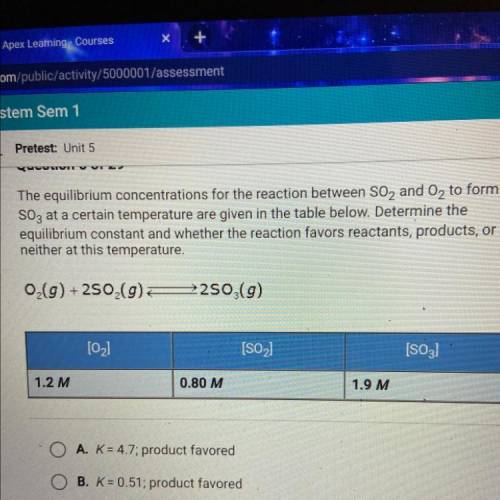
The equilibrium concentrations for the reaction between SO2 and O2 to form
SO3 at a certain temperature are given in the table below. Determine the
equilibrium constant and whether the reaction favors reactants, products, or
neither at this temperature.
O(g) +250 (9)
2250 (9)
[02]
[SO2)
[S03)
1.2 M
0.80 M
1.9 M
A. K = 4.7; product favored
B. K = 0.51; product favored
C. K = 0.51; reactant favored
o
D. K= 4.7; reactant favored

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1
You know the right answer?
The equilibrium concentrations for the reaction between SO2 and O2 to form
SO3 at a certain tempera...
Questions

Mathematics, 18.09.2020 04:01

Mathematics, 18.09.2020 05:01

History, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Health, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

English, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01



