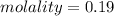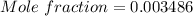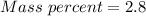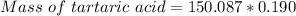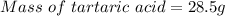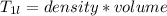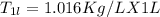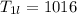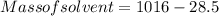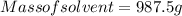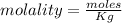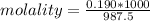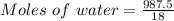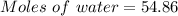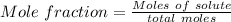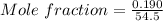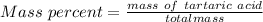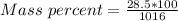Chemistry, 16.07.2021 01:00 911wgarcia
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If the beverage is 0.190 tartaric acid, what is the molal concentration? What is the mole fraction of tartaric acid and water? Calculate the mass percent of tartaric acid. The density of the solution is 1.016g/mL.

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 00:30
Arrange the elements in order of increasing electronegativity. use the periodic table to you arrange the elements. p o k mg
Answers: 2

Chemistry, 23.06.2019 06:00
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes? or no? this question is worth 20 points! let it be correct!
Answers: 1

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 3

Chemistry, 23.06.2019 16:00
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a) 3.4 mol h2so4 b) 6.8 mol h2so4 c) 10.2 mol h2so4 d) 13.6 mol h2so4 a) 3.4 mol h2so4
Answers: 1
You know the right answer?
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If...
Questions


Mathematics, 28.01.2021 03:20


Business, 28.01.2021 03:20


Chemistry, 28.01.2021 03:20


Mathematics, 28.01.2021 03:20




English, 28.01.2021 03:20


Mathematics, 28.01.2021 03:20



Mathematics, 28.01.2021 03:20

Mathematics, 28.01.2021 03:20

Arts, 28.01.2021 03:20

Physics, 28.01.2021 03:20