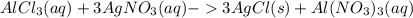Chemistry, 12.07.2021 19:30 swaggyd6603
If 13.50 mL of an aluminum chloride solution is needed to reach the equivalence point with 10.00 mL of 0.109 M silver nitrate solution, determine the molarity of the aluminum chloride solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 23.06.2019 03:30
27 drag each label to the correct location on the image. a particular exosolar system has five planets in total: a, b, c, d, and e. the table lists the orbital periods of these planets in days. planet orbital period (days) a 600 b 80 c 1,000 d 500 e 100 move each planet to its orbit in the system.
Answers: 3

Chemistry, 23.06.2019 09:20
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of this solution? use the periodic table in the toolbar if needed. 0.0625 g what mass of naoh must be added to the solution to increase the concentration to 30.00% by mass? g
Answers: 2

Chemistry, 23.06.2019 10:20
Determine the mass of the object below with accuracy and to the correct degree of precision. a. 324.2 g b. 324 g c. 324.30 g d. 324.25 g
Answers: 3
You know the right answer?
If 13.50 mL of an aluminum chloride solution is needed to reach the equivalence point with 10.00 mL...
Questions

Biology, 27.06.2019 23:30

Mathematics, 27.06.2019 23:30

Mathematics, 27.06.2019 23:30

Mathematics, 27.06.2019 23:30


Mathematics, 27.06.2019 23:30

History, 27.06.2019 23:30


History, 27.06.2019 23:30


Mathematics, 27.06.2019 23:30


Mathematics, 27.06.2019 23:30

Mathematics, 27.06.2019 23:30


History, 27.06.2019 23:30


Biology, 27.06.2019 23:30