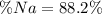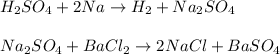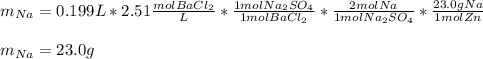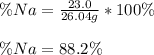Chemistry, 10.07.2021 02:00 jetblackcap
A 26.04 g mixture of zinc and sodium is reacted with a stoichiometric amount of sulfuric acid. The reaction mixture is then reacted with 199 mL of 2.51 M barium chloride to produce the maximum possible amount of barium sulfate. Determine the percent sodium by mass in the original mixture.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 10:00
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3
You know the right answer?
A 26.04 g mixture of zinc and sodium is reacted with a stoichiometric amount of sulfuric acid. The r...
Questions

History, 13.04.2021 01:50



Mathematics, 13.04.2021 01:50



Geography, 13.04.2021 01:50




Mathematics, 13.04.2021 01:50








English, 13.04.2021 01:50