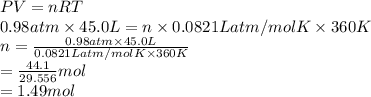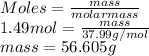Chemistry, 09.07.2021 20:20 kaygirlnelson6258
Answer the following question:
Calculate the mass of 45.0 L of F2 at 87.0° C and 750. mm Hg.
Use the Ideal Gas Law formula and here are values for R:
8.134 (L * kPa)/(mol * K)
0.0821 (atm * L)/(mol * K)
62.364 (L * mmHg)/(mol * K)
Include the following with your
Which Gas Law constant did you use or which "R" value did you use? Why?
The numerical answer to the question.
An explanation of the correct number of significant figures you will use for the numerical answer.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 19:30
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Answer the following question:
Calculate the mass of 45.0 L of F2 at 87.0° C and 750. mm Hg.
<...
<...
Questions


Chemistry, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00


Mathematics, 19.10.2021 14:00


Chemistry, 19.10.2021 14:00




Social Studies, 19.10.2021 14:00


Mathematics, 19.10.2021 14:00

History, 19.10.2021 14:00

Biology, 19.10.2021 14:00

History, 19.10.2021 14:00

English, 19.10.2021 14:00

English, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00

 at 87.0° C and 750 mm Hg is 56.605 g.
at 87.0° C and 750 mm Hg is 56.605 g.  = (87.0 + 273) K = 360 K
= (87.0 + 273) K = 360 K