
Chemistry, 09.07.2021 03:20 yakshp3702
During a reaction, ΔH for reactants is −750 kJ/mol and ΔH for products is 920 kJ/mol. Which statement is correct about the reaction? (5 points)
Group of answer choices
It is endothermic because the energy required to break bonds in the reactants is less than the energy released when the products are formed.
It is endothermic because the energy required to break bonds in the reactants is greater than the energy released when the products are formed.
It is exothermic because the energy required to break bonds in the reactants is less than the energy released when the products are formed.
It is exothermic because the energy required to break bonds in the reactants is greater than the energy released when the products are formed.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

You know the right answer?
During a reaction, ΔH for reactants is −750 kJ/mol and ΔH for products is 920 kJ/mol. Which statemen...
Questions

Mathematics, 24.03.2021 06:10

Mathematics, 24.03.2021 06:10


Chemistry, 24.03.2021 06:10






Mathematics, 24.03.2021 06:10

Mathematics, 24.03.2021 06:10

Mathematics, 24.03.2021 06:10

English, 24.03.2021 06:10



Mathematics, 24.03.2021 06:10



Health, 24.03.2021 06:10

Mathematics, 24.03.2021 06:10

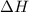 is always negative.
is always negative.

