
A chemist fills a reaction vessel with 9.47 atm nitrogen monoxide (NO) gas, 2.61 atm chlorine (C12) gas, and 8.64 atm nitrosyl chloride (NOCI) gas at a temperature of 25.0°C. Under these conditions, calculate the reaction free energy AG for the following chemical reaction:
2NO(g) + Cl2(g) = 2NOCI (g)
Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3


Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

Chemistry, 23.06.2019 07:00
Determine the length of the object shown. 97.8 mm 97.80 mm 97 mm 98 mm
Answers: 1
You know the right answer?
A chemist fills a reaction vessel with 9.47 atm nitrogen monoxide (NO) gas, 2.61 atm chlorine (C12)...
Questions


History, 11.07.2019 23:00

Biology, 11.07.2019 23:00

Social Studies, 11.07.2019 23:00






Mathematics, 11.07.2019 23:00

English, 11.07.2019 23:00


Business, 11.07.2019 23:00

Mathematics, 11.07.2019 23:00

Mathematics, 11.07.2019 23:00

Biology, 11.07.2019 23:00

Computers and Technology, 11.07.2019 23:00

English, 11.07.2019 23:00


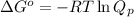
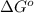 = Gibbs free energy change
= Gibbs free energy change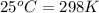
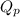 = reaction quotient =
= reaction quotient = 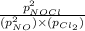
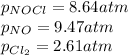
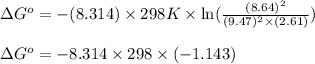
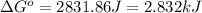 (Conversion factor: 1 kJ = 1000 J)
(Conversion factor: 1 kJ = 1000 J)

