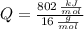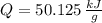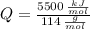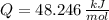Cuando se quema 1 mol de metano –o sea, 16 g–, se desprenden 802
kJ/mol.
○ Cuando se quema 1 mol de octano –o sea, 114 g–, se desprenden 5500
kJ/mol.
Pareciera que el octano puede brindar más energía al quemarse, pero vamos a
hacer un análisis más cuidadoso. Comparemos la combustión de igual masa de
cada combustible.
● Calculen la cantidad de calor que se desprende cuando se quema un gramo
de cada combustible.
1 gr de metano aporta……………………………
1 gr de octano aporta………………………………

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is a scientific theory? a. a scientist's guess about how something works b. the results of an experiment obtained using the scientific method c. a proven fact that will never change d. an idea that is backed by data from many sources
Answers: 2

Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2


Chemistry, 22.06.2019 18:00
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1
You know the right answer?
Cuando se quema 1 mol de metano –o sea, 16 g–, se desprenden 802
kJ/mol.
○ Cuando se quema 1...
○ Cuando se quema 1...
Questions

Biology, 21.08.2019 09:30


History, 21.08.2019 09:30



History, 21.08.2019 09:30

Social Studies, 21.08.2019 09:30


Mathematics, 21.08.2019 09:30

Social Studies, 21.08.2019 09:30


Social Studies, 21.08.2019 09:30


Mathematics, 21.08.2019 09:30



Mathematics, 21.08.2019 09:30

History, 21.08.2019 09:30


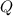 ), en kilojoules por mol, es igual a la cantidad de energía liberada por mol de compuesto (
), en kilojoules por mol, es igual a la cantidad de energía liberada por mol de compuesto (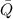 ), en kilojoules por mol, dividido por su masa molar (
), en kilojoules por mol, dividido por su masa molar (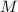 ), en gramos por mol:
), en gramos por mol: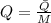 (1)
(1)