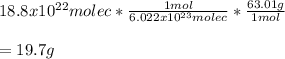Chemistry, 08.07.2021 04:40 reneebrown017
A sample of hydrogen nitrate or nitric acid, HNO 3 contains 18.8 x 1022 molecules. How much mass of nitric acid are in the sample?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2

Chemistry, 23.06.2019 07:00
Agas has an empirical formula ch4. 0.16g of the gas occupies a volume of 240cm^3 what is the molecular formula of the me anyone who !
Answers: 1

Chemistry, 23.06.2019 09:00
A0.10 m aqueous solution of sodium sulfate is a better conductor of electricity than a 0.10 m aqueous solution of sodium chloride. which of the following best explains this observation? (a) sodium sulfate is more soluble in water than sodium chloride. (b) sodium sulfate has a higher molar mass than sodium chloride. (c) to prepare a given volume of 0.10 m solution, the mass of sodium sulfate needed is more than twice the mass of sodium chloride needed. (d) more moles of ions are present in a given volume of 0.10 m sodium sulfate than in the same volume of 0.10 m sodium chloride. (e) the degree of dissociation of sodium sulfate in solution is significantly greater than that of sodium chloride.
Answers: 2
You know the right answer?
A sample of hydrogen nitrate or nitric acid, HNO 3 contains 18.8 x 1022 molecules.
How much mass of...
Questions

Mathematics, 14.04.2021 22:00



English, 14.04.2021 22:00

Social Studies, 14.04.2021 22:00



Mathematics, 14.04.2021 22:00


Mathematics, 14.04.2021 22:00

Biology, 14.04.2021 22:00


Business, 14.04.2021 22:00

English, 14.04.2021 22:00




Mathematics, 14.04.2021 22:00