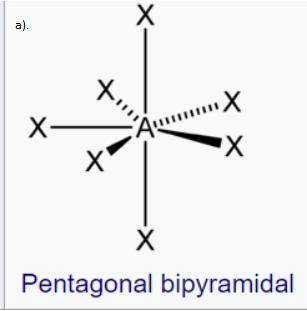
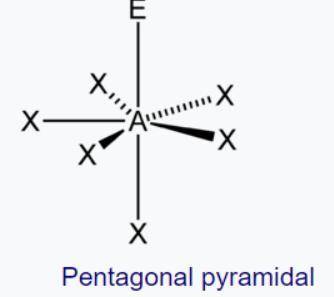
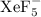The geometry is called pentagonal bipyramidal. This has a lot of similarities to a 6-coordinate molecule except there are 5 domains in one plane. Predict the following based on this information:
a. Draw the structure of a molecule with 7 bonding domains
b. List all the bond angles possible between adjacent atoms
c. Predict the electron and molecule geometry for a molecule with 6 bonding domains and a single lone pair
d. Predict the electron and molecule geometry for a molecule with 5 bonding domains and two lone pairs

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
In which direction will the following reaction go if the standard reduction potentials are 0.80 v for ag/ag+ and –0.44 v for fe/fe2+? ag+ + fe → ag + fe2+ a.)forward b.)the reaction cannot occur. c.) not enough information is given. d.) reverse
Answers: 1

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1


Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1
You know the right answer?
The geometry is called pentagonal bipyramidal. This has a lot of similarities to a 6-coordinate mole...
Questions

Mathematics, 12.03.2021 21:30





Biology, 12.03.2021 21:30



History, 12.03.2021 21:30


Mathematics, 12.03.2021 21:30

Chemistry, 12.03.2021 21:30

Mathematics, 12.03.2021 21:30

English, 12.03.2021 21:30


Mathematics, 12.03.2021 21:30

History, 12.03.2021 21:30

Social Studies, 12.03.2021 21:30

Spanish, 12.03.2021 21:30

Mathematics, 12.03.2021 21:30

 .
. 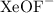 .
.