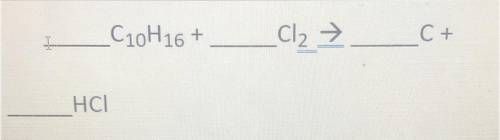C20H16+
_Cl2 →
C+
HCl
Please help me fast
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2
You know the right answer?
Questions



Business, 28.09.2019 07:00

History, 28.09.2019 07:00




Mathematics, 28.09.2019 07:00

Social Studies, 28.09.2019 07:00


Health, 28.09.2019 07:00

Mathematics, 28.09.2019 07:00

Biology, 28.09.2019 07:00


History, 28.09.2019 07:00

Physics, 28.09.2019 07:00


Mathematics, 28.09.2019 07:00


Social Studies, 28.09.2019 07:00