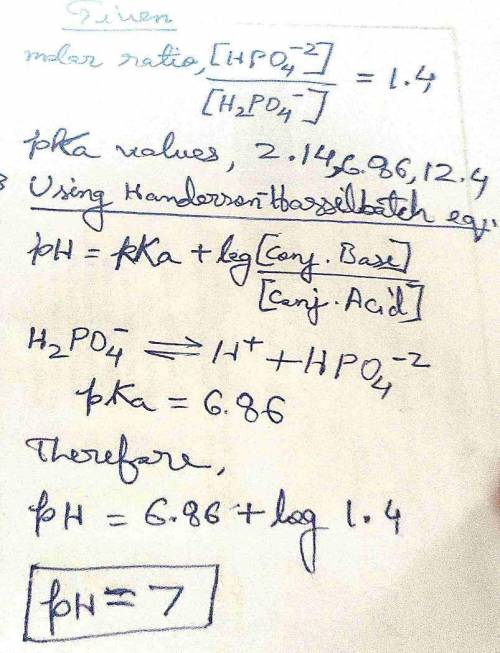Chemistry, 03.07.2021 01:20 charlesiarenee0
The molar ratio of HPO42- to H2PO4- in a solution is 1.4. Calculate the pH of the solution. Phosphoric acid (H3PO4) is a triprotic acid with 3 pKa values: 2.14, 6.86, and 12.4.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1
You know the right answer?
The molar ratio of HPO42- to H2PO4- in a solution is 1.4. Calculate the pH of the solution. Phosphor...
Questions

Mathematics, 22.02.2021 07:40




Mathematics, 22.02.2021 07:40





Chemistry, 22.02.2021 07:40

Computers and Technology, 22.02.2021 07:40


Mathematics, 22.02.2021 07:40

Mathematics, 22.02.2021 07:40


Mathematics, 22.02.2021 07:40