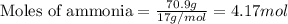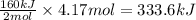2NH3(g)→N2(g)+3H2(g)

Chemistry, 01.07.2021 15:40 rowdycar313p0ao5k
A chemist measures the energy change
ΔH during the following reaction:
2NH3(g)→N2(g)+3H2(g)
ΔH=160kJUse the information to answer the following questions. This reaction is:.
a. endothermic
b. exothermic
Suppose 70.9 g of NH3 react. Will any heat be released or absorbed?
a. Yes, absorbed
b. Yes, released
c. No.
If you said heat will be released or absorbed in the second part of this question, calculate how much heat will be released or absorbed. Round your answer to 3 significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
If the root word engage means “to connect with something,” what does the word disengage mean in the following sentence? he disengaged the gears by stepping on the clutch pedal.a.added more engine powerb.activated a connection to the pedalc.stalled the engined.released a connection to the pedal
Answers: 1

Chemistry, 22.06.2019 00:30
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1


Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
A chemist measures the energy change
ΔH during the following reaction:
2NH3(g)→N2(g)+3H2(g)
2NH3(g)→N2(g)+3H2(g)
Questions






Mathematics, 20.09.2019 19:10







Mathematics, 20.09.2019 19:10




History, 20.09.2019 19:10


Mathematics, 20.09.2019 19:10

Mathematics, 20.09.2019 19:10

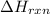 is positive for these reactions.
is positive for these reactions.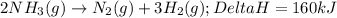
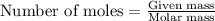 ......(1)
......(1)