
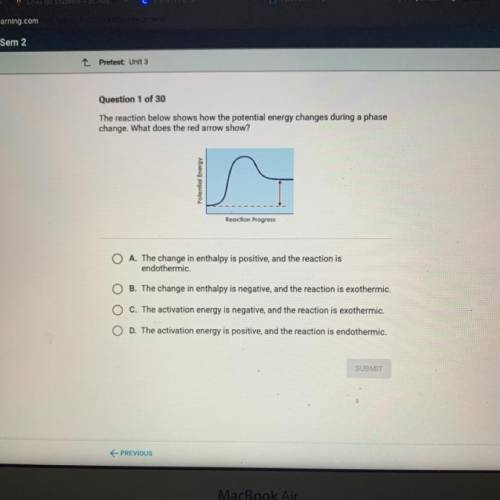
The reaction below shows how the potential energy changes during a phase
change. What does the red arrow show?
Potential Energy
Reaction Progress
A. The change in enthalpy is positive, and the reaction is
endothermic.
B. The change in enthalpy is negative, and the reaction is exothermic.
C. The activation energy is negative, and the reaction is exothermic.
D. The activation energy is positive, and the reaction is endothermic.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2


You know the right answer?
The reaction below shows how the potential energy changes during a phase
change. What does the red...
Questions


Social Studies, 22.07.2019 10:30

Chemistry, 22.07.2019 10:30

History, 22.07.2019 10:30


Chemistry, 22.07.2019 10:30

History, 22.07.2019 10:30

Biology, 22.07.2019 10:30

Spanish, 22.07.2019 10:30


Social Studies, 22.07.2019 10:30

Social Studies, 22.07.2019 10:30


Social Studies, 22.07.2019 10:30

Mathematics, 22.07.2019 10:30

Mathematics, 22.07.2019 10:30

History, 22.07.2019 10:30





