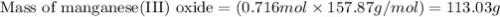Using the balanced equation below,
how many grams of manganese(III)
oxide would be produced f...

Chemistry, 30.06.2021 07:20 brittanyelliott028
Using the balanced equation below,
how many grams of manganese(III)
oxide would be produced from the
complete reaction of 46.8 g of zinc?
Zn + 2MnO2 + H20 — Zn(OH)2 + Mn203


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
Questions





Mathematics, 12.09.2019 23:30


Mathematics, 12.09.2019 23:30

Mathematics, 12.09.2019 23:30


English, 12.09.2019 23:30

Mathematics, 12.09.2019 23:30

Mathematics, 12.09.2019 23:30

Biology, 12.09.2019 23:30

Physics, 12.09.2019 23:30

Chemistry, 12.09.2019 23:30

History, 12.09.2019 23:30

Computers and Technology, 12.09.2019 23:30



Biology, 12.09.2019 23:30

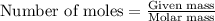 ......(1)
......(1)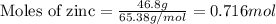
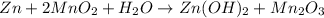
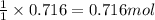 of manganese(III) oxide
of manganese(III) oxide