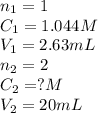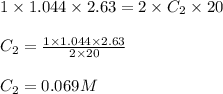Chemistry, 28.06.2021 18:40 summerjoiner
Standardization of a Borax solution (Na2B4O7). You are given a 1.044 M solution of H2SO4. It takes 2.63 mL of this H2SO4 to reach the end point. Knowing it takes 1 H2SO4 to neutralize 2 Na2B4O7, what was the concentration of this Borax solution?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1


Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1
You know the right answer?
Standardization of a Borax solution (Na2B4O7). You are given a 1.044 M solution of H2SO4. It takes 2...
Questions

Mathematics, 14.09.2020 02:01

History, 14.09.2020 02:01

History, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 02:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

English, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

Mathematics, 14.09.2020 03:01

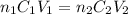 ....(1)
....(1)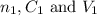 are the n-factor, concentration and volume of sulfuric acid
are the n-factor, concentration and volume of sulfuric acid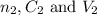 are the n-factor, concentration and volume of borax solution.
are the n-factor, concentration and volume of borax solution.