
Chemistry, 28.06.2021 15:40 coolgirl10020031
An experiment was performed under identical conditions as yours. The absorbance of the penny solution was recorded as 0.219 absorbance units. A calibration plot of absorbance vs. concentration of Cu(II) (M) yielded the following trendline equation:
y= 11.589x - 0.0002
Required:
What is the concentration (mol/L) of the penny solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
An experiment was performed under identical conditions as yours. The absorbance of the penny solutio...
Questions


Mathematics, 18.02.2021 22:30

Mathematics, 18.02.2021 22:30



Social Studies, 18.02.2021 22:30




English, 18.02.2021 22:30

Mathematics, 18.02.2021 22:30

Mathematics, 18.02.2021 22:30



Mathematics, 18.02.2021 22:30

Mathematics, 18.02.2021 22:30

English, 18.02.2021 22:30

Chemistry, 18.02.2021 22:30


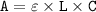 --- (1)
--- (1)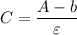 ----(3)
----(3)

