
Chemistry, 25.06.2021 05:40 genyjoannerubiera
For each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S of the system, decrease S, or leave S unchanged. If you don’t have enough information to decide, check the “not enough information “button in the last column.
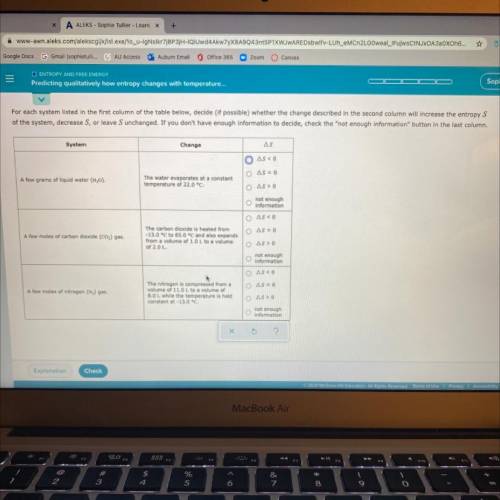

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Diamond, graphite, and fullerenes share what property? a. they are all made of carbon (c) bonded to a metal. b. their shape. c. they are all made of carbon (c). d. they are all good conductors.
Answers: 1

Chemistry, 21.06.2019 16:00
Review the branily terms and services guides well u know what i never did so go have a nice ice cream sunday
Answers: 1

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
For each system listed in the first column of the table below, decide (if possible) whether the chan...
Questions


Physics, 26.09.2019 09:30


Mathematics, 26.09.2019 09:30


English, 26.09.2019 09:30


History, 26.09.2019 09:30

Mathematics, 26.09.2019 09:30


Mathematics, 26.09.2019 09:30


Mathematics, 26.09.2019 09:30





English, 26.09.2019 09:30


Biology, 26.09.2019 09:30



