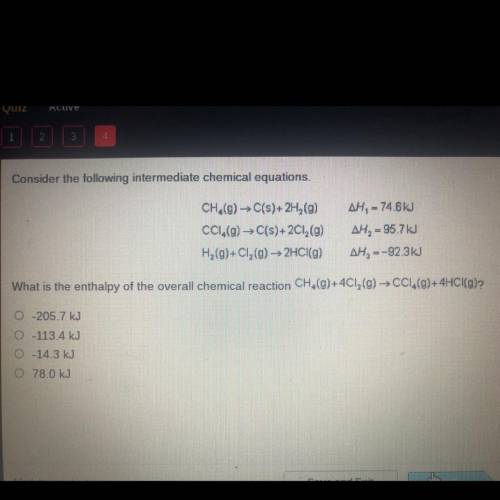
Consider the following intermediate chemical equations.
CH.(g) → C(s)+2H (9)
CC1.(g) → C(s)+2...

Chemistry, 23.06.2021 19:50 nataliajaquez02
Consider the following intermediate chemical equations.
CH.(g) → C(s)+2H (9)
CC1.(g) → C(s)+2Cl2(g)
H2(g)+C1, (g) → 2HCl(g)
AH, = 74.6 kJ
AH, = 95.7 kJ
AH, =-92.3kJ
What is the enthalpy of the overall chemical reaction CH,(g)+4C12(g) → CC1,(9)+4HCI(g)?
O-205.7 kJ
0-113.4 kJ
-14.3 kJ
0 78.0 kJ

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3
You know the right answer?
Questions






Social Studies, 21.02.2021 20:30

English, 21.02.2021 20:30


Mathematics, 21.02.2021 20:30

History, 21.02.2021 20:30

Mathematics, 21.02.2021 20:30

Mathematics, 21.02.2021 20:30

Spanish, 21.02.2021 20:30

Biology, 21.02.2021 20:30



Spanish, 21.02.2021 20:30

Mathematics, 21.02.2021 20:30

English, 21.02.2021 20:30

Spanish, 21.02.2021 20:30



