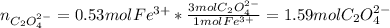Chemistry, 23.06.2021 07:40 smithmalyk4
Kc is 1.67 x 10^20 at 25 °C for the formation of iron(III) oxalate complex ion:
Fe^3+ (aq) + 3C2O4^2- (aq) <--> [Fe(C204)3]^3- (aq)
Determine the number of moles of C2O4^2- used to react with 0.53 moles of Fe^3+.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 23.06.2019 03:30
The molar mass of iron(fe) is 55.8 g/mol. what is the mass in grams of 2.25 moles of iron?
Answers: 1

Chemistry, 23.06.2019 09:20
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1
You know the right answer?
Kc is 1.67 x 10^20 at 25 °C for the formation of iron(III) oxalate complex ion:
Fe^3+ (aq) + 3C2O4^...
Questions


Mathematics, 16.01.2020 10:31



Mathematics, 16.01.2020 10:31

English, 16.01.2020 10:31

Mathematics, 16.01.2020 10:31





Mathematics, 16.01.2020 10:31



English, 16.01.2020 10:31


Physics, 16.01.2020 10:31


Mathematics, 16.01.2020 10:31

Social Studies, 16.01.2020 10:31

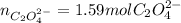
![Kc=\frac{[[Fe(C_2O_4)_3]^{3-}]}{[Fe^{3+}][C_2O_4^{2-}]^3}](/tpl/images/1382/0865/5bd65.png)