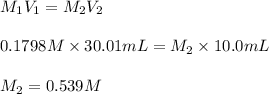g Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with a sodium hydroxide solution according to the equation H C 2 H 3 O 2 + N a O H ⟶ H 2 O + N a C 2 H 3 O 2 If you require 30.01 mL of 0.1798 M N a O H solution to titrate 10.0 mL of H C 2 H 3 O 2 solution, what is the molar concentration of acetic acid in the vinegar? Type

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2
You know the right answer?
g Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with...
Questions

History, 20.08.2019 10:30



Mathematics, 20.08.2019 10:30

Mathematics, 20.08.2019 10:30

Mathematics, 20.08.2019 10:30

History, 20.08.2019 10:30

Physics, 20.08.2019 10:30




Biology, 20.08.2019 10:30

History, 20.08.2019 10:30

Mathematics, 20.08.2019 10:30



History, 20.08.2019 10:30


Mathematics, 20.08.2019 10:30

Social Studies, 20.08.2019 10:30

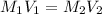
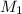 and
and 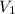 are the concentration and volume of base.
are the concentration and volume of base.
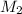 and
and 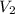 are the concentration and volume of an acid.
are the concentration and volume of an acid.