
Chemistry, 22.06.2021 18:00 keiracoles
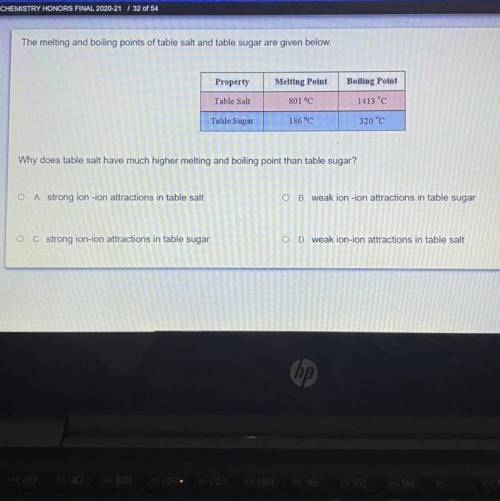
The melting and boiling points of table salt and table sugar are given below.
Why does table salt have much higher melting and boiling point than table sugar?
o A strong ion -ion attractions in table salt
O B. weak ion-ion attractions in table sugar
O C. strong ion-ion attractions in table sugar
O
D. weak ion-ion attractions in table salt

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 23:00
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1
You know the right answer?
The melting and boiling points of table salt and table sugar are given below.
Why does table salt h...
Questions

Mathematics, 17.06.2021 03:40


Mathematics, 17.06.2021 03:40

Mathematics, 17.06.2021 03:40

Mathematics, 17.06.2021 03:40

Chemistry, 17.06.2021 03:40


English, 17.06.2021 03:40


Mathematics, 17.06.2021 03:40

Mathematics, 17.06.2021 03:40

Mathematics, 17.06.2021 03:40

Social Studies, 17.06.2021 03:40


Mathematics, 17.06.2021 03:40


Mathematics, 17.06.2021 03:40


Mathematics, 17.06.2021 03:40

Mathematics, 17.06.2021 03:40



