
Chemistry, 22.06.2021 04:40 bearminar2156
2 NH3 + 3 CuO --> 3 Cu + N2 + 3 H2O
In the above equation, how many moles of N2 can be made when 113.6 grams of CuO are consumed?
Round your answer to the nearest tenth. If your answer is a whole number like 4, report the answer as 4.0
Use the following molar masses. If you do not use these masses, the computer will mark your answer incorrect. :
Element Molar Mass
Hydrogen
1
Nitrogen
14
Copper
63.5
Oxygen
16

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3

Chemistry, 21.06.2019 21:00
Solar energy is energy from the sun that is converted into thermal or energy. a. nuclear b. mechanical c. electrical d. chemical
Answers: 2

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
You know the right answer?
2 NH3 + 3 CuO --> 3 Cu + N2 + 3 H2O
In the above equation, how many moles of N2 can be made when...
Questions

Computers and Technology, 19.04.2020 22:44



Mathematics, 19.04.2020 22:44





Mathematics, 19.04.2020 22:44

Mathematics, 19.04.2020 22:44


Chemistry, 19.04.2020 22:44

History, 19.04.2020 22:44


Mathematics, 19.04.2020 22:44

Biology, 19.04.2020 22:44



Chemistry, 19.04.2020 22:45

Mathematics, 19.04.2020 22:45

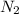 are produced in the reaction
are produced in the reaction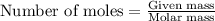 ......(1)
......(1)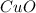 = 113.6 g
= 113.6 g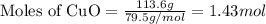
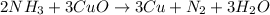
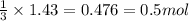 of
of 

