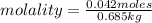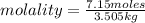Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
Calculate the molality of each of the following solutions:
(a) 14.3 g of sucrose (C12H22O11) in 685...
Questions

Mathematics, 17.10.2020 23:01





Mathematics, 17.10.2020 23:01

Biology, 17.10.2020 23:01

Physics, 17.10.2020 23:01


English, 17.10.2020 23:01


Physics, 17.10.2020 23:01





English, 17.10.2020 23:01



Mathematics, 17.10.2020 23:01

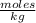
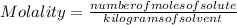
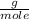 , then 14.3 grams of the compound represents the following number of moles:
, then 14.3 grams of the compound represents the following number of moles: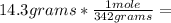 0.042 moles
0.042 moles