
Chemistry, 19.06.2021 04:20 zanaplen27
PLEASE HELP QUICK
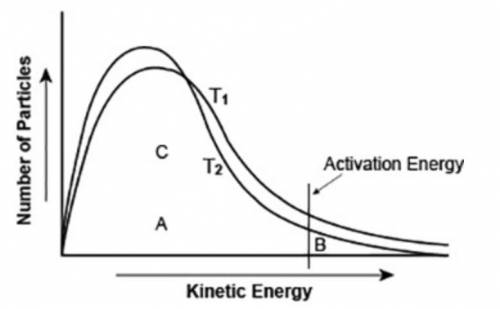
The graph shows the distribution of energy in the particles of two gas samples at different temperatures, T1 and T2. A, B, and C represent individual particles.
Based on the graph, which of the following statements is likely to be true? (3 points)
Particle A is more likely to participate in the reaction than particle B.
Particle C is more likely to participate in the reaction than particle B.
The number of particles able to undergo a chemical reaction is less than the number that is not able to.
More gas particles participate in the reaction at T2 than at T1.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2


Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
PLEASE HELP QUICK
The graph shows the distribution of energy in the particles of two gas samples at...
Questions


History, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30



Mathematics, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

Physics, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

History, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

History, 27.06.2019 18:30

English, 27.06.2019 18:30


Mathematics, 27.06.2019 18:30

Biology, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30

Mathematics, 27.06.2019 18:30



