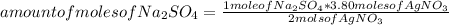Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4 (aq) --> Ag2SO4 (s) 2 NaNO3 (aq) If 3.80 moles of AgNO3 and 3.05 moles of Na2SO4 are present initially, how many moles of excess reactant will remain after precipitation is complete

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

You know the right answer?
Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4...
Questions


Biology, 16.12.2020 21:10


Mathematics, 16.12.2020 21:10

Computers and Technology, 16.12.2020 21:10

Biology, 16.12.2020 21:10

Mathematics, 16.12.2020 21:10




Mathematics, 16.12.2020 21:10

English, 16.12.2020 21:10

Mathematics, 16.12.2020 21:10

Mathematics, 16.12.2020 21:10



Spanish, 16.12.2020 21:10

Mathematics, 16.12.2020 21:10


Mathematics, 16.12.2020 21:10