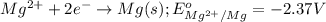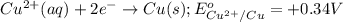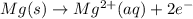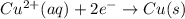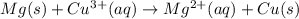Chemistry, 18.06.2021 06:10 angscott9638
I neeed help nowww HELP NOOW
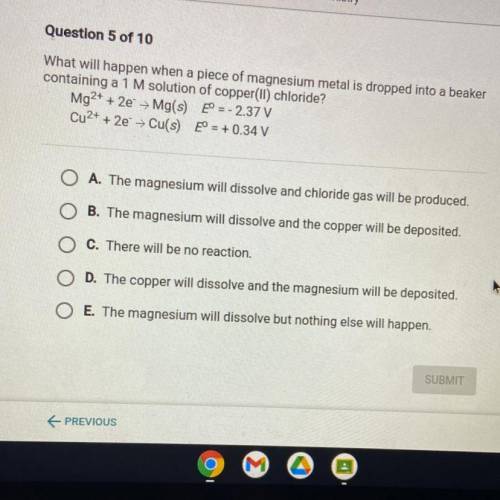
What will happen when a piece of magnesium metal is dropped into a beaker
containing a 1 M solution of copper(II) chloride?
Mg2+ + 2e + Mg(s) eº = -2.37 V
Cu2+ + 2e → Cu(s) E° = + 0.34 V

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1


Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2
You know the right answer?
I neeed help nowww HELP NOOW
What will happen when a piece of magnesium metal is dropped into a bea...
Questions




Mathematics, 09.03.2020 01:22

Mathematics, 09.03.2020 01:22

Mathematics, 09.03.2020 01:22


Chemistry, 09.03.2020 01:23


Mathematics, 09.03.2020 01:24

Mathematics, 09.03.2020 01:25

Social Studies, 09.03.2020 01:25




Mathematics, 09.03.2020 01:27

Mathematics, 09.03.2020 01:27

Social Studies, 09.03.2020 01:29