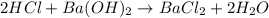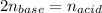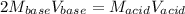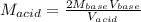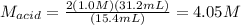Chemistry, 17.06.2021 18:20 zhellyyyyy
The molarity of an aqueous solution of hydrochloric acid, , is determined by titration with a M barium hydroxide, , solution. If 31.2 mL of barium hydroxide are required to neutralize 15.4 mL of the acid, what is the molarity of the hydrochloric acid solution?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1


Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
The molarity of an aqueous solution of hydrochloric acid, , is determined by titration with a M bari...
Questions





Mathematics, 11.10.2019 01:30




Mathematics, 11.10.2019 01:30


Social Studies, 11.10.2019 01:30

Mathematics, 11.10.2019 01:30

History, 11.10.2019 01:30


English, 11.10.2019 01:30

Biology, 11.10.2019 01:30

Biology, 11.10.2019 01:30


History, 11.10.2019 01:30