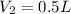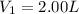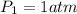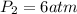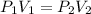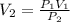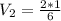Chemistry, 17.06.2021 02:50 live4dramaoy0yf9
2.00 liters of gas at 1.00 atm and conpresssed it to a pressure of 6.00 atm, what would the volume of the gas be

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
2.00 liters of gas at 1.00 atm and conpresssed it to a pressure of 6.00 atm, what would the volume o...
Questions

Mathematics, 13.07.2019 10:30

Physics, 13.07.2019 10:30


History, 13.07.2019 10:30


Chemistry, 13.07.2019 10:30

Biology, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30



Biology, 13.07.2019 10:30

English, 13.07.2019 10:30


Physics, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30

English, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30

Mathematics, 13.07.2019 10:30