.
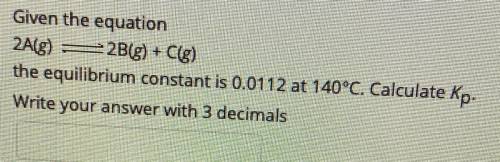
Given the equation
2A(g) - 2B(g) + C(g)
the equilibrium constant is 0.0112 at 140°C....

Chemistry, 16.06.2021 22:20 emilyphillips1681
.
Given the equation
2A(g) - 2B(g) + C(g)
the equilibrium constant is 0.0112 at 140°C. Calculate Kp.
Write your answer with 3 decimals

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
Questions





Social Studies, 17.05.2021 20:00

Mathematics, 17.05.2021 20:00


Mathematics, 17.05.2021 20:00



Mathematics, 17.05.2021 20:00

Business, 17.05.2021 20:00



Business, 17.05.2021 20:00







