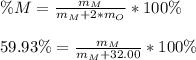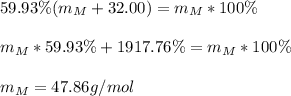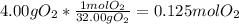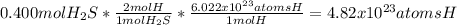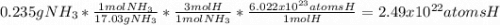Chemistry, 16.06.2021 21:40 milessims3953
A metal, M, forms an oxide having the formula MO2 containing 59.93% metal by mass. Determine the atomic weight in g/mole of the metal (M). Please provide your answer in 2 decimal places.
a. How many moles of oxygen atoms are found in 4.00 g of O2?
b. How many atoms of hydrogen are found in 0.400 mol of H2S?
c. How many hydrogen atomsare present in 0.235 g of NH3?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
A metal, M, forms an oxide having the formula MO2 containing 59.93% metal by mass. Determine the ato...
Questions

Physics, 04.03.2021 19:10

English, 04.03.2021 19:10


Mathematics, 04.03.2021 19:10

French, 04.03.2021 19:10

Social Studies, 04.03.2021 19:10

Mathematics, 04.03.2021 19:10


Physics, 04.03.2021 19:10

French, 04.03.2021 19:10

Mathematics, 04.03.2021 19:10





Mathematics, 04.03.2021 19:10


Mathematics, 04.03.2021 19:10

Arts, 04.03.2021 19:10

Chemistry, 04.03.2021 19:10