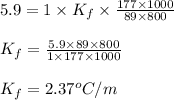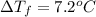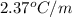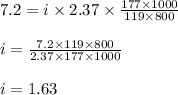Chemistry, 16.06.2021 15:40 PersonPerson13260
When of alanine are dissolved in of a certain mystery liquid , the freezing point of the solution is less than the freezing point of pure . Calculate the mass of potassium bromide that must be dissolved in the same mass of to produce the same depression in freezing point. The van't Hoff factor for potassium bromide in .

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1
You know the right answer?
When of alanine are dissolved in of a certain mystery liquid , the freezing point of the solution is...
Questions

Mathematics, 29.01.2021 02:00


Mathematics, 29.01.2021 02:00

Mathematics, 29.01.2021 02:00

English, 29.01.2021 02:00




Computers and Technology, 29.01.2021 02:00

Mathematics, 29.01.2021 02:00

Mathematics, 29.01.2021 02:00

Biology, 29.01.2021 02:00

Computers and Technology, 29.01.2021 02:00


Mathematics, 29.01.2021 02:00




Mathematics, 29.01.2021 02:00

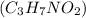 are dissolved in 800.0 g of a certain mystery liquid X, the freezing point of the solution is
are dissolved in 800.0 g of a certain mystery liquid X, the freezing point of the solution is 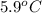 lower than the freezing point of pure X. On the other hand, when 177.0 g of potassium bromide are dissolved in the same mass of X, the freezing point of the solution is
lower than the freezing point of pure X. On the other hand, when 177.0 g of potassium bromide are dissolved in the same mass of X, the freezing point of the solution is 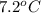 lower than the freezing point of pure X. Calculate the van't Hoff factor for potassium bromide in X.
lower than the freezing point of pure X. Calculate the van't Hoff factor for potassium bromide in X.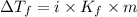
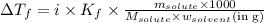 ......(1)
......(1)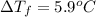
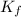 = freezing point depression constant
= freezing point depression constant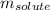 = Given mass of solute (alanine) = 177. g
= Given mass of solute (alanine) = 177. g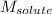 = Molar mass of solute (alanine) = 89 g/mol
= Molar mass of solute (alanine) = 89 g/mol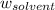 = Mass of solvent = 800.0 g
= Mass of solvent = 800.0 g