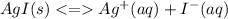Chemistry, 14.06.2021 21:00 jungkookie001
Compare the solubility of silver iodide in each of the following aqueous solutions:
a. 0.10 M AgCH3COO
b. 0.10 M NaI
c. 0.10 M KCH3COO
d. 0.10 M NH4NO3
1. More soluble than in pure water.
2. Similar solubility as in pure water.
3. Less soluble than in pure water.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Clouds form when water vapor to form small droplets. a. humidifies b. condenses c. evaporates d. precipitates
Answers: 2

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2
You know the right answer?
Compare the solubility of silver iodide in each of the following aqueous solutions:
a. 0.10 M AgCH3...
Questions

History, 03.07.2019 05:30

English, 03.07.2019 05:30





Biology, 03.07.2019 05:30

Biology, 03.07.2019 05:30

Chemistry, 03.07.2019 05:30



History, 03.07.2019 05:30

Social Studies, 03.07.2019 05:30

Biology, 03.07.2019 05:30

Biology, 03.07.2019 05:30

Biology, 03.07.2019 05:30



Chemistry, 03.07.2019 05:30

Mathematics, 03.07.2019 05:30