
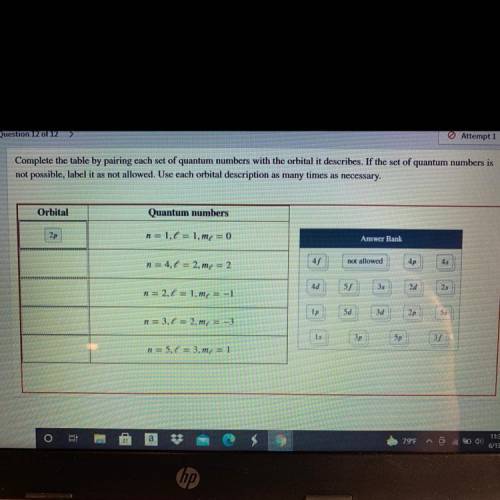
Complete the table by pairing each set of quantum numbers with the orbital it describes. If the set of quantum numbers is
not possible, label it as not allowed. Use each orbital description as many times as necessary.
Orbital
Quantum numbers
2p
n = 1.6 = 1.me = 0
Answer Bank
n = 4.1 = 2.m = 2
45
not allowed
4p
45
4d
n = 2.1 = 1.mx = -1
55
35
2d
25
Ip
5d
3d
2p
55
n = 3,6 = 2.mx = -3
1s
3p
5p
35
n = 5.0 = 3.m4 = 1
7
O
Bi
11:36 PM

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

You know the right answer?
Complete the table by pairing each set of quantum numbers with the orbital it describes. If the set...
Questions




Computers and Technology, 03.01.2022 18:00

Advanced Placement (AP), 03.01.2022 18:00

Mathematics, 03.01.2022 18:00


German, 03.01.2022 18:00

Mathematics, 03.01.2022 18:10

History, 03.01.2022 18:10


Computers and Technology, 03.01.2022 18:10


Chemistry, 03.01.2022 18:10




Social Studies, 03.01.2022 18:10

Mathematics, 03.01.2022 18:10



